|
ESTER
(ESS-tuhr)
Any
one of a group of organic compounds that are formed by the
reaction between an alcohol and an acid
Common
clues:
Organic compound; Perfume ingredient; Aromatic compound; Banana
oil, for one; Chemical
compound; Fragrant compound; Any acetate chemically
Crossword
puzzle frequency:
5 times a year
Frequency
in English language:
39571 / 86800
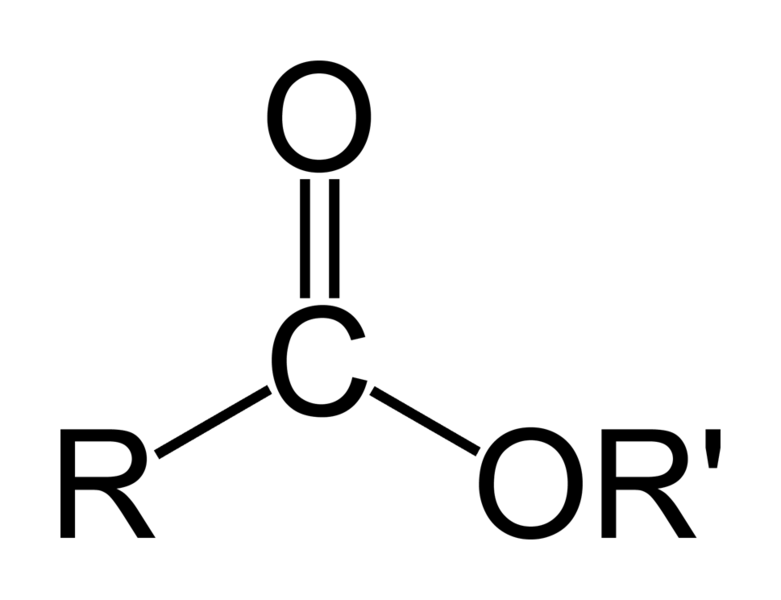
In
chemistry, esters are organic compounds in which an organic group
(symbolized by R' in this article) replaces a hydrogen atom (or
more than one) in an oxygen acid. An oxygen acid is an acid whose
molecule has an -OH group from which the hydrogen (H) can
dissociate as an H+ ion.
Esters
participate in hydrogen bonds as hydrogen-bond acceptors, but
cannot act as hydrogen-bond donors, unlike their parent alcohols.
This ability to participate in hydrogen bonding makes them more
water-soluble than their parent hydrocarbons. However, the
limitations on their hydrogen bonding also make them more
hydrophobic than either their parent alcohols or parent acids.
Their lack of hydrogen-bond-donating ability means that ester
molecules cannot hydrogen-bond to each other, which makes esters
generally more volatile than a carboxylic acid of similar
molecular weight. This property makes them very useful in organic
analytical chemistry: unknown organic acids with low volatility
can often be esterified into a volatile ester, which can then be
analysed using gas chromatography, gas liquid chromatography, or
mass spectrometry. Many esters have distinctive odors, which has
led to their use as artificial flavorings and fragrances. For
example:
allyl
hexanoate pineapple
benzyl
acetate pear, strawberry, jasmine
bornyl
acetate pine tree flavor
butyl
butyrate pineapple
ethyl
acetate peach, pineapple, raspberry
ethyl
butyrate banana, pineapple, strawberry
This
article is licensed under the GNU
Free Documentation License.
It uses material from the Wikipedia
article "Ester".
9
Tu >1 07 Moroccan
|